Rational understanding of the catalytic mechanism of molybdenum carbide in polysulfide conversion in lithium–sulfur batteries†
Abstract
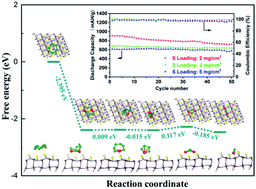
Lithium–sulfur (Li–S) batteries are promising candidates for next-generation energy storage devices due to their high theoretical energy density and whose practical applications are mainly hampered by the shuttle effect of intermediate polysulfides (LiPSs). Anchoring materials, such as β-Mo2C, with strong chemical interaction has been proposed to improve the electrochemical performance of Li–S batteries. However, the chemical bonding and conversion reaction of LiPSs on the Mo2C surface are not well studied. Here, we report on the discovery that the superior performance of Mo2C originates from the sulfur termination. By combining X-ray spectroscopy measurements and theoretical calculations, we reveal that sulfur can passivate the Mo2C (101) surface, which not only offers moderate chemical interaction with LiPSs but also facilitates the conversion reactions during both the discharge and charge processes. Our results suggest that it is important to consider the sulfurization of catalysts with metal surfaces when they are used to accelerate the conversion of polysulfides.


 Please wait while we load your content...
Please wait while we load your content...