Direct utilization of air and water as feedstocks in the photo-driven nitrogen reduction reaction over a ternary Z-scheme SiW9Co3/PDA/BWO hetero-junction†
Abstract
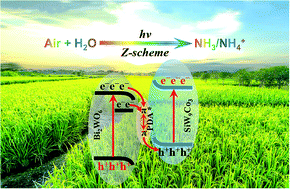
Sustainable solar-to-ammonia conversion under ambient conditions using air and water as feedstocks is a promising approach to supply low-concentrated ammonia (fertilizer) for farms. In the present work, the semiconductor-like polyoxometalate, tricobalt substituted silicotungstate [α-SiW9O37Co3(H2O)3]10− (SiW9Co3), is stabilized on a Bi2WO6 (BWO) substrate via a poly-dopamine (PDA) adhesive to construct a ternary SiW9Co3/PDA/BWO composite. In the Z-scheme SiW9Co3/PDA/BWO composite, the migration of photo-generated charger carriers between SiW9Co3 and BWO, with a narrow band gap, is facilitated by π–π* electronic delocalization of PDA. The catalytic efficiency of ternary SiW9Co3/PDA/BWO is 6.0 times higher than that of pristine BWO, owing to multiple chemisorptions of molecular nitrogen, the presence of oxygen vacancies, and efficient separation and migration of photo-generated electron–hole pairs. A quantitative reactivity–structure relationship is established according to the correlation between transient photocurrent response and catalytic efficiency. The enhancement factor of the Z-scheme mechanism is estimated to be 1.7–2.5. Essentially, the photo-driven nitrogen reduction reaction is carried out in pure water under both pure nitrogen and air atmospheres with visible light (and simulated sunlight) irradiation. Direct utilization of air and water as feedstocks highlights its promising prospects in marching towards a “solar ammonia refinery”, where the photo-driven nitrogen reduction devices are built near a farm to produce fertilizer as needed.


 Please wait while we load your content...
Please wait while we load your content...