A mechanistic study of mesoporous TiO2 nanoparticle negative electrode materials with varying crystallinity for lithium ion batteries†
Abstract
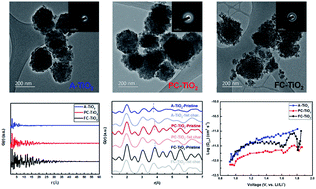
Nanoscale oxide-based negative electrodes are of great interest for lithium ion batteries due to their high energy density, power density and enhanced safety. In this work, we conducted a case study on mesoporous TiO2 nanoparticle negative electrodes with uniform size and varying crystallinity in order to investigate the trend in the electrochemical properties of oxide-based nanoscale negative electrodes with varying crystallinity. Mesoporous solid spherical TiO2 nanoparticles with a uniform particle size and varying crystallinity, i.e., amorphous TiO2 (A-TiO2), partially crystalline TiO2 (PC-TiO2) and fully crystalline TiO2 (FC-TiO2) nanoparticles were studied. At low current rate (quasi steady-state), the specific capacity of the samples drops with the decrease of crystallinity. Ex situ synchrotron pair distribution function analysis reveals that the 1D zigzag Li ion diffusion pathway becomes expanded with the increase of crystallinity, which promotes ion mobility and charge storage. At high current rates (away from equilibrium states), however, the A-TiO2 sample demonstrates slightly larger capacity than the FC-TiO2 sample, both of which show larger capacities than that of the PC-TiO2 sample. Both A-TiO2 and FC-TiO2 samples exhibit higher capacitive contribution to the charge storage and larger Li+ diffusivity than those of the PC-TiO2 sample, which explains their better rate capability. Moreover, the larger Li+ diffusivity of the A-TiO2 sample leads to the slightly larger specific capacity than the FC-TiO2 sample at the highest current rate.


 Please wait while we load your content...
Please wait while we load your content...