Trade-off between oxygen reduction reaction activity and CO2 stability in a cation doped Ba0.9Co0.7Fe0.3O3−δ perovskite cathode for solid oxide fuel cells†
Abstract
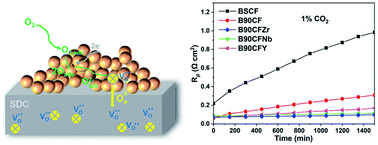
The excellent oxygen permeability of the perovskite BaCoO3 and its applicability as a cathode in solid oxide fuel cells are hindered by stability issues of carbonate formation in the presence of CO2, which degrades its excellent oxygen surface-exchange kinetics during the oxygen reduction reaction (ORR) process. In this work, we present a new type of Ba0.9Co0.7Fe0.2M0.1O3−δ (M = Zr, Nb, Y) perovskite oxide with high CO2 tolerance with respect to their original compound Ba0.9Co0.7Fe0.3O3−δ (B90CF) by doping cations at the B-site. These cathodes show low polarization resistances (Rp) of 0.0687, 0.0719, and 0.0853 Ω cm2 for Ba0.9Co0.7Fe0.2Y0.1O3−δ (B90CFY), Ba0.9Co0.7Fe0.2Zr0.1O3−δ (B90CFZr), and Ba0.9Co0.7Fe0.2Nb0.1O3−δ (B90CFNb) at 700 °C, respectively, which are comparable to B90CF (0.0655 Ω cm2). Moreover, after exposure to air containing 1% CO2 for 1500 min, the electrochemical impedance spectroscopy (EIS) test demonstrated the improved CO2 tolerance for the B90CFM cathode with Zr (Nb, Y) doping, where the Rp of B90CFZr only increased by 31%, ∼1/10 of that for B90CF. Barium carbonate on the B90CFZr cathode was obviously suppressed compared to B90CF according to SEM combined with EDS, XRD and FT-TR analysis. Doping high-valence metals at the B site of the B90CFM perovskite may increase the acidity on the surface and metal-oxygen bond free energy (〈ABE〉), leading to enhanced resistance to CO2. The trade-off between electrocatalytic activity and stability enable B90CFZr to be a promising cathode material for low and medium temperature SOFCs.


 Please wait while we load your content...
Please wait while we load your content...