Characterization of a new rechargeable Zn/PVA-KOH/Bi2O3 battery: structural changes of the Bi2O3 electrode†
Abstract
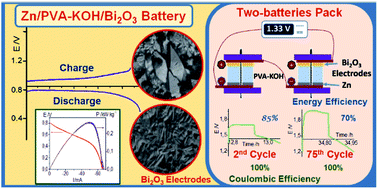
In this study, bismuth oxide is utilized as positive electrode in a rechargeable Zn-based battery using a gel polymer electrolyte. Bi2O3 is reduced to Bi metal during the discharge process of the Zn/PVA-KOH/Bi2O3 battery, obtaining efficiencies higher than 90% from the Bi2O3 mass initially used for discharge currents ≤10 mA. During the charge process, Bi metal is oxidized to recover the Bi2O3. A great number of charge/discharge cycles are performed for a single battery and for two batteries connected in series. This battery develops coulombic efficiencies of 100% for more than 100 cycles and energy efficiencies higher than 80% after 80 cycles. XRD, AES and XPS measurements demonstrate the formation of Bi metal during the discharge and the build-up of Bi2O3 structure during the charge. Besides, SEM and EDX techniques confirm the structural changes observed during the reduction and oxidation processes. Finally, discharge and charge mechanisms of the Bi2O3 electrode tested on a Zn/PVA-KOH/Bi2O3 battery are discussed.
- This article is part of the themed collection: Sustainable Energy & Fuels Cover Art


 Please wait while we load your content...
Please wait while we load your content...