The simplest Diels–Alder reactions are not endo-selective†
Abstract
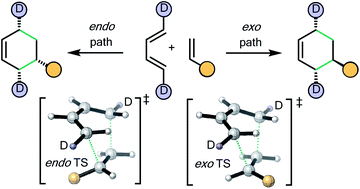
There is a widespread perception that the high level of endo selectivity witnessed in many Diels–Alder reactions is an intrinsic feature of the transformation. In contrast to expectations based upon this existing belief, the first experimental Diels–Alder reactions of a novel, deuterium-labeled 1,3-butadiene with commonly used mono-substituted alkenic dienophiles (acrolein, methyl vinyl ketone, acrylic acid, methyl acrylate, acrylamide and acrylonitrile) reveal kinetic endo : exo ratios close to 1 : 1. Maleonitrile, butenolide, α-methylene γ-butyrolactone, and N-methylmaleimide behave differently, as does methyl vinyl ketone under Lewis acid catalysis. CBS-QB3 calculations incorporating solvent and temperature parameters give endo : exo product ratios that are in near quantitative agreement with these and earlier experimental findings. This work challenges the preconception of innate endo-selectivity by providing the first experimental evidence that the simplest Diels–Alder reactions are not endo-selective. Trends in behaviour are traced to steric and electronic effects in Diels–Alder transition structures, giving new insights into these fundamental processes.


 Please wait while we load your content...
Please wait while we load your content...