A modular and divergent approach to spirocyclic pyrrolidines†
Abstract
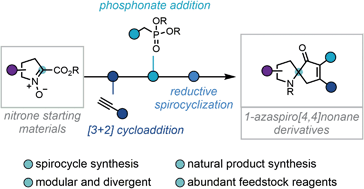
An efficient three-step sequence to afford a valuable class of spirocyclic pyrrolidines is reported. A reductive cleavage/Horner–Wadsworth–Emmons cascade facilitates the spirocyclisation of a range of isoxazolines bearing a distal β-ketophosphonate. The spirocyclisation precursors are elaborated in a facile and modular fashion, via a [3 + 2]-cycloaddition followed by the condensation of a phosphonate ester, introducing multiple points of divergence. The synthetic utility of this protocol has been demonstrated in the synthesis of a broad family of 1-azaspiro[4,4]nonanes and in a concise formal synthesis of the natural product (±)-cephalotaxine.
- This article is part of the themed collection: 2020 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...