Competition between N and O: use of diazine N-oxides as a test case for the Marcus theory rationale for ambident reactivity†
Abstract
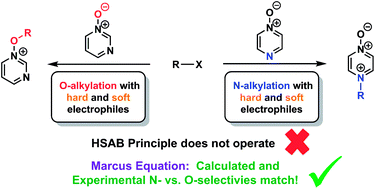
The preferred site of alkylation of diazine N-oxides by representative hard and soft alkylating agents was established conclusively using the 1H–15N HMBC NMR technique in combination with other NMR spectroscopic methods. Alkylation of pyrazine N-oxides (1 and 2) occurs preferentially on nitrogen regardless of the alkylating agent employed, while O-methylation of pyrimidine N-oxide (3) is favoured in its reaction with MeOTf. As these outcomes cannot be explained in the context of the hard/soft acid/base (HSAB) principle, we have instead turned to Marcus theory to rationalise these results. Marcus intrinsic barriers (ΔG‡0) and ΔrG° values were calculated at the DLPNO-CCSD(T)/def2-TZVPPD/SMD//M06-2X-D3/6-311+G(d,p)/SMD level of theory for methylation reactions of 1 and 3 by MeI and MeOTf, and used to derive Gibbs energies of activation (ΔG‡) for the processes of N- and O-methylation, respectively. These values, as well as those derived directly from the DFT calculations, closely reproduce the observed experimental N- vs. O-alkylation selectivities for methylation reactions of 1 and 3, indicating that Marcus theory can be used in a semi-quantitative manner to understand how the activation barriers for these reactions are constructed. It was found that N-alkylation of 1 is favoured due to the dominant contribution of ΔrG° to the activation barrier in this case, while O-alkylation of 3 is favoured due to the dominant contribution of the intrinsic barrier (ΔG‡0) for this process. These results are of profound significance in understanding the outcomes of reactions of ambident reactants in general.


 Please wait while we load your content...
Please wait while we load your content...