From reactive carbenes to chiral polyether macrocycles in two steps – synthesis and applications made easy?
Abstract
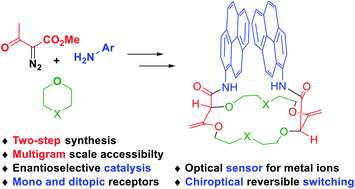
Chiral polyether macrocycles are versatile molecules. For their preparation, original two-step procedures were recently developed and present the advantages of high concentration conditions and simple starting reagents (stable diazo reagents, small cyclic ethers, aliphatic or aromatic amines). Enantiopure materials are readily afforded by CSP-HPLC on a semi-preparative scale. Flexibility and adaptability in the macrocyclic design are provided by a large selection of amines to choose from while the ring size and chemical nature are controlled by the choice of 5 to 7-membered cyclic ether precursors. Such macrocycles have already been used as asymmetric catalysts, mono and ditopic receptors, fluorescent sensors and probes, and chiroptical reversible switches.
- This article is part of the themed collection: Celebrating 10 years of Chemical Science


 Please wait while we load your content...
Please wait while we load your content...