Enantiomerically enriched tetrahydropyridine allyl chlorides†
Abstract
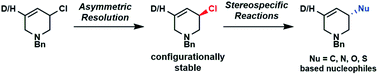
Enantiomerically enriched allyl halides are rare due to their configurational lability. Here we report stable piperidine-based allyl chloride enantiomers. These allyl chlorides can be produced via kinetic resolution, and undergo highly enantiospecific catalyst-free substitution reactions with C, N, O and S-based nucleophiles. DFT calculations and experiments with deuterium-labelled chloro-tetrahydropyridine, selectively prepared using H/D primary kinetic isotope effect, were used to investigate the mechanisms of resolution and substitution reactions. The allyl chlorides may also serve as valuable mechanistic tools for probing stereoselective reaction pathways.


 Please wait while we load your content...
Please wait while we load your content...