Mesoporous silica-encapsulated gold core–shell nanoparticles for active solvent-free benzyl alcohol oxidation†
Abstract
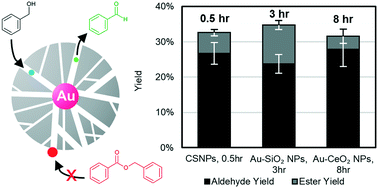
Silica-encapsulated gold core@shell nanoparticles (Au@SiO2 CSNPs) were synthesized via a tunable bottom-up procedure to catalyze the aerobic oxidation of benzyl alcohol. The nanoparticles exhibit a mesoporous shell which enhances selectivity by inhibiting the formation of larger species. Adding potassium carbonate to the reaction increased conversion from 17.3 to 60.4% while decreasing selectivity from 98.4 to 75.0%. A gold nanoparticle control catalyst with a similar gold surface area took 6 times as long to reach the same conversion, achieving only 49.4% selectivity. These results suggest that the pore size distribution within the inert silica shell of Au@SiO2 CSNPs inhibits the formation of undesired products to facilitate the selective oxidation of benzaldehyde despite a basic environment. A smaller activation energy, mass transport analysis, and mesopore distribution together suggest the Au@SiO2 CSNP catalyst demonstrates higher activity through beneficial in-pore orientation, promoting a lower activation energy mechanistic pathway. Taken together, this is a promising catalytic structure to optimize oxidation chemistries, without leveraging surface-interacting factors like chelating agents or active support surfaces.


 Please wait while we load your content...
Please wait while we load your content...