Highly efficient and selective membrane separation of copper from nickel in ammoniacal solution using mixtures of M5640 and BESO as membrane carriers
Abstract
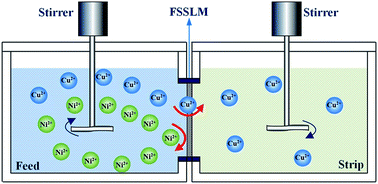
Separation of copper from nickel in ammoniacal/ammonium chloride solution using a flat-sheet supported liquid membrane impregnated with mixtures of Acorga M5640 and bis(2-ethylhexyl)sulfoxide (BESO) was investigated. The crucial parameters influencing copper transport and separation abilities of copper and nickel, such as carrier concentration of M5640 and BESO in the membrane phase, initial concentration of ions in the feed phase, H2SO4 concentration in the strip phase and membrane stability, were discussed. The results show that the mixtures of carriers (20 vol% M5640 + 20 vol% BESO) in the membrane have a considerable antagonistic effect on membrane transport of nickel, but favor copper transport. Nearly all of the copper was transferred from the feed phase to the strip phase after 12 hours with a flux of 2.05 × 10−5 mol m−2 s−1 under the following conditions: 100 mg L−1 each of the copper and nickel dissolved in 1.0 mol L−1 each of ammonia and ammonium chloride solution as the feed phase, 60 g L−1 H2SO4 as the strip phase, and stirring speed of 800 rpm in two aqueous phases. Meanwhile less than 3.8% of the nickel was transported into the strip phase over the same time. Copper and nickel were efficiently separated with a calculated factor of 26.3. Furthermore, satisfactory membrane stability was obtained with at least ten cycle runs in this separation system.


 Please wait while we load your content...
Please wait while we load your content...