Surface reduction properties of ceria–zirconia solid solutions: a first-principles study†
Abstract
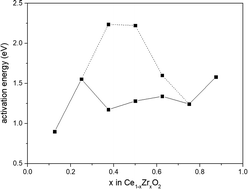
Based on the density functional theory (DFT), the reduction properties of Ce1−xZrxO2 (110) surfaces were systematically calculated using CO as a probe for thermodynamic study, and a large supercell was applied to build the whole composition range (x = 0.125, 0.250, 0.375, 0.500, 0.625, 0.750, 0.875). From the calculated energy barriers of CO oxidation by lattice oxygen, we found that composition Ce0.875Zr0.125O2 exhibited the most promising catalytic effectiveness with the lowest activation energy of 0.899 eV. Moreover, the active surface O3c ions coordinated by two Zr ions and one Ce ion were facilely released from their bulk positions than the O3c ions surrounded by two Ce ions and one Zr ion on Ce0.625Zr0.375O2, Ce0.500Zr0.500O2, and Ce0.375Zr0.625O2 (110) surfaces. This difference could be explained by the binding strength of O3c with different neighboring cations.


 Please wait while we load your content...
Please wait while we load your content...