Palladium-catalyzed double carbonylation of propargyl amines and aryl halides to access 1-aroyl-3-aryl-1,5-dihydro-2H-pyrrol-2-ones†
Abstract
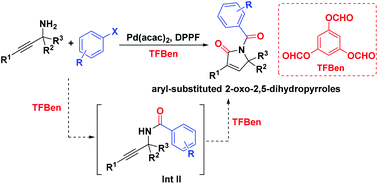
In this communication, we disclosed a palladium-catalyzed carbonylative procedure for the synthesis of 1-aroyl-3-aryl-1,5-dihydro-2H-pyrrol-2-ones from propargyl amines and aryl halides. By using TFBen as the CO source, the reaction provides a facile and straightforward access to various aryl-substituted 2-oxo-2,5-dihydropyrroles in good yields. The reaction mechanism is proven to be attractive as well.


 Please wait while we load your content...
Please wait while we load your content...