Controlled oxygen vacancy engineering on In2O3−x/CeO2−y nanotubes for highly selective and efficient electrocatalytic nitrogen reduction†
Abstract
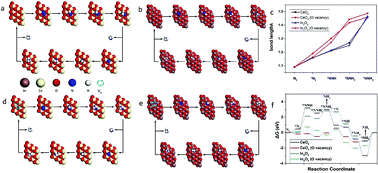
Introducing and adjusting the oxygen vacancies (VO) of transition metal oxides has been proposed as a significant and effective way to tackle the sluggish nitrogen reduction reaction (NRR) in the electrocatalysis process. In this work, we present the synthesis of an In2O3−x/CeO2−y nanotube with abundant VOvia the electrospinning technique, followed by the controlled post-treatment calcination process. It is demonstrated that the electrochemical properties of In2O3−x/CeO2−y are successfully optimized through VO engineering and the reaction mechanism of the NRR is the dissociative pathway, which remarkably enhanced electronic conduction and the NRR reaction kinetics simultaneously. The optimized In2O3−x/CeO2−y nanotube exhibits excellent NRR catalytic activity and durability, with an outstanding average yield of 26.1 μg h−1 mgcat−1 while the faradaic current efficiency of 16.1% at −0.3 V is ultrahigh versus the reversible hydrogen electrode. This study offers insights into designing excellent catalysts for the NRR through a combination of the nanotube structure and the adjustment of the VO strategy.


 Please wait while we load your content...
Please wait while we load your content...