Palladium-catalysed regio- and stereoselective arylative substitution of γ,δ-epoxy-α,β-unsaturated esters and amides by sodium tetraaryl borates†
Abstract
Palladium-catalysed reactions of γ,δ-epoxy-α,β-unsaturated esters and amides with NaBAr4 reagents proceeded regio- and stereoselectively, producing allylic homoallyl alcohols with aryl-substituents in the allylic position for a wide range of substrates. AsPh3 was found to be a competent ligand for the arylation reaction, whereas phosphine ligand/Lewis acidic organoboron combinations favoured the substitution reaction by oxygen nucleophiles (e.g. H2O, ROH).
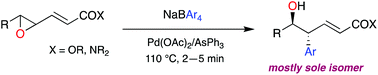
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...