Multiple targeting strategies achieve novel protein drug delivery into proapoptosis lung cancer cells by precisely inhibiting survivin†
Abstract
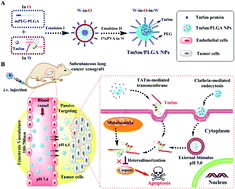
Therapeutic recombinant proteins have numerous advantages and benefits over chemical drugs, particularly high specificity and good biocompatibility. However, the therapeutic potential and clinical application of current anticancer protein drugs are limited as most biomarkers are located within cells, and multiple physiological barriers exist between the point of administration and the intracellular biomarker. Herein, we report a novel strategy to accurately deliver a cell-permeable dominant-negative TATm-Survivin (TmSm) protein (T34A) to intracellular survivin in cancer cells by overcoming multiple barriers in vivo. A poly(D,L-lactide-co-glycolide) (PLGA) inner core, a polyethylene glycol (PEG) modification, and a TATm peptide were simultaneously introduced to mediate tumor tissue targeting and response to pH-triggered TmSm release. Compared to free TmSm, the PEGylated-PLGA nanoparticle platform achieved a significantly higher cellular uptake efficiency (1.79-fold for A549 and 1.77-fold for Capan-2), effectively decreased IC50 (1.22-fold for A549 and 1.17-fold for Capan-2), and largely elevated apoptosis in different cancer cells (1.17-fold for A549 and 1.15-fold for Capan-2). Besides, this newly developed nanoplatform showed increased protein drug accumulation in the tumor site in A549-bearing nude mice and reached a tumor inhibition rate of 55.81% (1.35-fold versus free TmSm) by reducing the expression of intracellular survivin. All these results confirmed that our newly developed delivery strategy is a very promising tool, which helps protein drugs to cross multiple barriers in vivo and achieves precise targeting to intracellular biomarkers. This strategy could also be applied to other types of protein drugs to further improve their clinical anticancer therapeutic efficacy.


 Please wait while we load your content...
Please wait while we load your content...