Template-directed synthesis of Sm2Ti2O7 nanoparticles: a FRET-based fluorescent chemosensor for the fast and selective determination of picric acid
Abstract
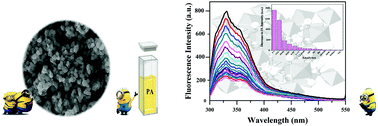
Due to security and health issues the detection of trace levels of nitro-explosives in solutions is of great importance. In the present study, Sm2Ti2O7 pyrochlore fluorescent nanoparticles were synthesized via a surfactant-directed method. A sol–gel processing route assisted by pluronic P123 in water was used for the synthesis of Sm2Ti2O7 pyrochlore fluorescent nanoparticles. The synthesized Sm2Ti2O7 nanoparticles were applied as a quenching-based fluorescent nanoprobe for the sensitive detection of trace levels of nitrate explosive chemicals, including 2,4,6-trinitrophenol (picric acid (PA)), 2,4,6-trinitrotoluene (TNT), 1,3,5-trinitro-1,3,5-triazinane (RDX) and 1,3,5,7-tetranitro-1,3,5,7-tetrazoctane (HMX). The fluorescence quenching behaviors of Sm2Ti2O7 were checked in acetonitrile and acetonitrile/water mixture media with excitation and emission maxima at 280 and 330 nm, respectively. The pyrochlore nanoprobe presents a more effective turn-off response for nitroaromatic explosives, especially for PA, with a detection limit of 0.01 nM and a high Stern–Volmer constant of 9.9 × 105 M−1 in acetonitrile solvent. Experimental studies such as time-resolved fluorescence decay measurements, comparison of the energy levels of the probe and analytes and analysis of the spectral overlap between the nanoprobe and analytes revealed that the possible mechanism for this dynamic probe-quenching is the fluorescence resonance energy transfer (FRET). The proposed nanoprobe introduces a quick, reproducible, and cost-effective sensing system for the sensitive detection of nitro-explosives in solution with the highest selectivity to PA. The chemical inertness and photostability of the present STO probe give it the opportunity to be stored for a long time and be used for the monitoring of nitro-explosive pollutants in industrial effluents.


 Please wait while we load your content...
Please wait while we load your content...