Half-sandwich scandium dibenzyl complexes bearing penta- or tetra-arylcyclopentadienyl ligands: synthesis, structure and syndiospecific styrene polymerization activity†
Abstract
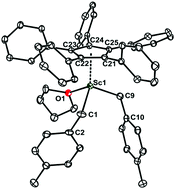
A series of half-sandwich scandium dibenzyl complexes has been synthesized via a one-pot reaction of potassium derivatives of pentaarylcyclopentadienes (Cp(Ar*)5 or CpAr5) (Ar* = 3,5-tBu2–C6H3; Ar = 3,5-iPr2–C6H3) or tetraarylcyclopentadienes (Cp(Ar*)4H, CpAr4H and CpPh4H) with the cationic scandium dibenzyl complex [Sc(p-CH2C6H4-Me)2(THF)x][BPh4], generated in situ by the reaction of [Sc(p-CH2C6H4-Me)3(THF)2] with [NEt3H][BPh4] in THF. [(CpAr5)Sc(p-CH2C6H4-Me)2(THF)] (2) bearing a pentaarylcyclopentadienyl ligand displays two η1-coordinated benzyl ligands without any obvious interaction of the ipso-carbon of the phenyl groups. In comparison, the two benzyl ligands in [(CpPh4H)Sc(p-CH2C6H4-Me)2(THF)] (5) are supported by a tetraphenylcyclopentadienyl bond to the scandium center in two different patterns: one is η1-coordination and the other is the η2-coordination mode. In combination with [Ph3C][B(C6F5)4] and AliBu3, these complexes showed highly active syndiospecific polymerization of styrene, up to 587 (kg(sPS) mol(Sc)−1 h−1). The catalytic activity of these complexes decreased with the increasing steric hindrance of Cp ligands around the scandium centre in the order of CpPh4H > CpAr4H > Cp(Ar*)4H > CpAr5 > Cp(Ar*)5H.


 Please wait while we load your content...
Please wait while we load your content...