Computational optimization of a novel solid-state sensor for stable assay of isoxsuprine hydrochloride in the presence of its nephrotoxic/hepatotoxic photothermal degradation products: application in different sampling matrices†
Abstract
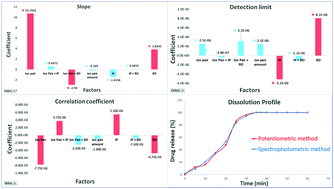
The pharmaceutical industry carries out analysis throughout the different stages of production to guarantee safety, potency, and purity. This work introduces a novel solid-state-potentiometric sensor for the selective assay of isoxsuprine hydrochloride (ISX) in the presence of its nephrotoxic and hepatotoxic photothermal degradation products. Preliminary studies specified the critical factors affecting the sensor performance. A two-level four-factor fractional factorial experimental design was constructed to evaluate the main effects and interactions among the specified factors. Factors included categorical factors (such as the ion-exchanger, binder, and ionophore), and numerical factors, such as the relative amounts of the matrix components. Eight sensors containing different levels of the studied factors were studied and outputs describing the sensor performance were estimated, such as the Nernstian slope, detection limit, and correlation coefficient. RStudio® software was used to calculate the prediction model used for the evidence-based selection of the optimum sensor composition. The optimized sensor proved a slope of 59.71 mV per decade, a limit of detection of 2.5 × 10−6 M, and a correlation coefficient approaching unity within the concentration range of 3.38 × 10−6–1 × 10−2 M. The sensor expressed a selective response to ISX in the presence of four nephrotoxic and hepatotoxic photothermal degradation products and common ionic contaminants. After ICH validation, the method proved its applicability throughout the different phases of pharmaceutical production (raw material, formulation, and biological samples). The fast and stable sensor response enabled the real-time inline dissolution testing of ISX tablets. The method was experimentally and statistically compared with the official USP dissolution method.


 Please wait while we load your content...
Please wait while we load your content...