The photophysical properties of cycloparaphenylene-based compounds with figure-eight configurations†
Abstract
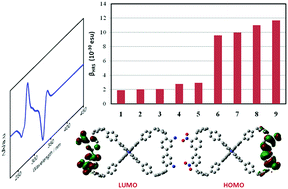
Cyclic conjugated compounds, especially those with inflection points, have become popular in chemistry and materials science as a result of their unique structures and interesting properties. Moreover, they demonstrate great potential in the development of high-performance optoelectronic and electronic devices. Understanding their photophysical properties at the atomic/electronic level is important for not only improving their performance but also extending their applicability. In this work, we investigated the photophysical properties of a series of conjugated macrocyclic compounds, which have figure-eight shapes and two inflections, with the aid of density functional theory (DFT) and time-dependent DFT (TDDFT) calculations. The parent compound, compound 1, exhibits a π → π* electron transition, which is located at the inflection point of the benzenes. In comparison with the parent compound as well as compounds with donors or acceptors, the introduction of both a donor and acceptor greatly reduces the band gap, modulates the HOMO/LUMO distributions, and modifies the electronic transition properties (e.g. the presence of an obvious intramolecular charge transfer). On the other hand, their second-order nonlinear optical (NLO) response values, ranging from 1.992 to 11.656 × 10−30 esu, are sensitive to the attributes of the substituted groups, demonstrating that these compounds are excellent candidates for second-order NLO materials.


 Please wait while we load your content...
Please wait while we load your content...