Crosslinking modification of a porous metal–organic framework (UIO-66) and hydrogen storage properties
Abstract
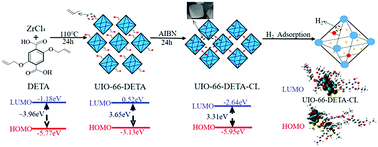
A crosslinkable UIO-66 type MOF material denoted UIO-66-DETA composed of Zr(IV) metal ions and 2,5-diallyl ether terephthalic acid (DETA) was synthesized, and then by initiating double bond crosslinking, a new crosslinking MOF material denoted UIO-66-DETA-CL was obtained. UIO-66-DETA and UIO-66-DETA-CL were characterized by FTIR spectroscopy, PXRD, 1H solid-state NMR spectroscopy, SEM, nitrogen sorption, thermogravimetric analysis and hydrogen storage tests to analyze their structure and properties. The results showed that a crosslinked structure has been formed, and UIO-66-DETA-CL can maintain its structure below 340 °C, compared with UIO-66-DETA; its carbon residual rate is improved, which shows that the crosslinking modification enhances the thermal stability. Meanwhile, the hydrogen uptake capacity of UIO-66-DETA-CL reaches 5.36 wt% at 77 K and 4 MPa. Furthermore, the hydrogen adsorption enthalpy of UIO-66-DETA-CL (ΔHUIO-66-DETA-CL) calculated using the Arrhenius equation reaches −0.889 kJ mol−1, which is significantly improved by crosslinking modification. The crosslinked structure changes the pore size and increases the active adsorption sites in organic ligands. The framework of UIO-66-DETA-CL can provide more polar charges to cause some hydrogen molecules to be negatively charged because of a small HOMO–LUMO gap, and a lower LUMO energy level of UIO-66-DETA-CL enables stronger negative charge acceptability. Therefore, crosslinking modification enhances the material's hydrogen storage capacity. This result provides a new idea for the crosslinking modification of MOFs.


 Please wait while we load your content...
Please wait while we load your content...