Synthesis of Ni2P/Al2O3 utilizing triphenylphosphine (TPP) as the phosphorus source for hydrodeoxygenation of benzofuran
Abstract
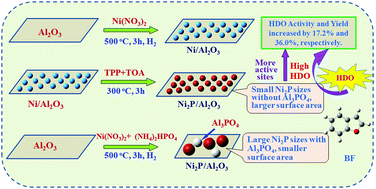
A novel route to synthesize highly active Ni2P/Al2O3 (TPP) utilizing triphenylphosphine (TPP) as the phosphorus source at a low temperature of 573 K is described. The as-prepared catalysts were characterized by X-ray diffraction (XRD), CO uptake, Brunner–Emmett–Teller (BET) measurements, and X-ray photoelectron spectroscopy (XPS). The catalytic activity of the Ni2P/Al2O3 (TPP) catalyst and the role of the TPP phosphorus source were studied using hydrodeoxygenation (HDO) of benzofuran (BF) as a probe reaction. The results show that the use of TPP as the phosphorus source could suppress the strong interaction between phosphate and Al2O3, thereby the formation of AlPO4 was avoided. As compared to the Ni2P/Al2O3 prepared by using (NH4)2HPO4 as the phosphorus source, Ni2P/Al2O3 (TPP) possessed significantly higher surface area and smaller Ni2P particle size. The HDO activity and yield of O-free products over the Ni2P/Al2O3 (TPP) catalyst were increased by 17.2% and 36.0%, respectively, when compared with those found for Ni2P/Al2O3 prepared using (NH4)2HPO4. The use of TPP as the phosphorus source could effectively promote the dehydration of 2-ethylphenol (2-EtPh) to form ethylbenzene (EB), and the demethylation of ethylcyclohexane (ECH) to methylcyclohexane (MCH).


 Please wait while we load your content...
Please wait while we load your content...