Inhibition and molecular mechanism of diosmetin against xanthine oxidase by multiple spectroscopies and molecular docking
Abstract
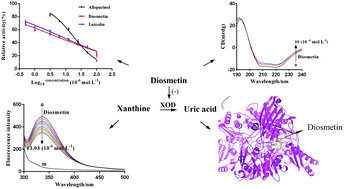
Diosmetin, as a methoxyflavone compound, was investigated for its interaction and mechanism with xanthine oxidase (XOD), which is a key enzyme catalyzing xanthine to uric acid and finally causing hyperuricemia. Using a kinetic analysis method, diosmetin was shown to possess a strong inhibitory activity for XOD in a reversibly competitive manner with an IC50 value of (5.08 ± 0.48) × 10−6 mol L−1. The results of fluorescence titrations showed that the combination of diosmetin with XOD gave spontaneous, exothermic static quenching, and hydrogen bonding and van der Waals forces were the main driving forces. It was suggested that diosmetin increased the polarity of the tyrosine and tryptophan microenvironment of XOD by synchronous fluorescence analysis. The results of ultraviolet absorption spectroscopy, circular dichroism and molecular docking showed that diosmetin could bind to the active site of XOD to form a new diosmetin–XOD complex. In these experiments, diosmetin reduced the content of α-helices, β-sheets and β-turns of XOD, which led to the instability of the XOD structure and the decrease of its catalytic function. Furthermore, the 4′-methoxy group of diosmetin had a higher affinity for XOD than the 4′-hydroxyl group of luteolin. To the best of our knowledge, this is the first comprehensive report to present the inhibitory activities of diosmetin against XOD.


 Please wait while we load your content...
Please wait while we load your content...