The presence of mixed-valent silver in the uranyl phenylenediphosphonate framework†
Abstract
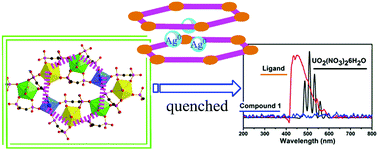
In this work, a new 2D layered silver uranyl phosphonate compound Ag01.51[AgI4(UO2)3(O3PC6H4PO3)(O3PC6H4PO3H)2] (denoted as compound 1) was synthesized by reaction of 1,2-phenylenediphosphonic acid (H4PDPA) with UO2(NO3)2·6H2O and AgNO3 under mild hydrothermal conditions. The uranyl layered framework consists of two types of UO8 and UO7 coordination units simultaneously connected by two kinds of phenylenediphosphonate linkers. To our surprise, the presence of both silver ions and metallic silver atoms in the free space between the layers is identified by the single crystal X-ray diffraction, EDS, and XPS techniques. The ratio of Ag+ and Ag0 was estimated to be 2.81 : 1, based on the primary peak area of Ag 3d5/2 from the XPS results, which corroborates with the ratio of 2.65 : 1 obtained from the XRD results. More importantly, the incorporation of the mixed-valent silver sites results in the quenching of the fluorescent emission of the uranyl phosphonate compound as evidenced by its single crystal fluorescence spectrum.


 Please wait while we load your content...
Please wait while we load your content...