1,3,4-Oxadiazole based thermostable energetic materials: synthesis and structure–property relationship†
Abstract
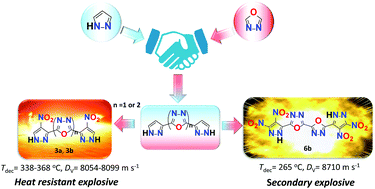
The increasing demands from both military and civilian applications inspired us to explore novel energetic materials with improved safety and energetic performance. Here, a class of C–C bond bridged polyazole energetic materials (based on 1,3,4-oxadiazole bridged polynitropyrazoles) were synthesized and fully characterized. A prediction model (two-dimensional (2D)-fingerprint and relevant Hirshfeld surfaces) was proposed to study the correlations between structures and physicochemical properties. Additionally, their energetic properties including detonation velocity, detonation pressure, sensitivity and thermal stability were fully characterized. Of these, compounds 3a and 3b possess excellent thermal stabilities (3a: Tdec = 338 °C and 3b: Tdec = 368 °C), desirable sensitivities (IS > 40 J, FS > 360 N) and higher energetic properties (3a: Dv = 8099 m s−1, P = 27.14 GPa and 3b: Dv = 8054 m s−1, P = 26.53 GPa) than hexanitrostilbene (HNS) (a typical heat resistant explosive: Tdec = 318 °C; Dv = 7612 m s−1, P = 24.30 GPa; IS = 5 J, FS = 240 N). Compound 6b exhibits high decomposition temperatures (Tdec = 265 °C), low sensitivities (IS = 30 J; FS = 360 N) and comparable detonation properties (Dv = 8710 m s−1, P = 32.75 GPa) to RDX. Considering their desirable energetic properties, they could be applied in different classes of energetic materials such as heat resistant explosives (3a, 3b) and secondary explosives (6b), respectively.


 Please wait while we load your content...
Please wait while we load your content...