A more sustainable and highly practicable synthesis of aliphatic isocyanides†
Abstract
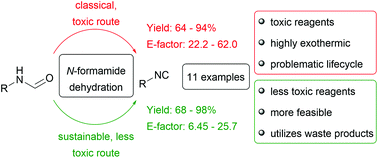
Synthesis protocols to convert N-formamides into isocyanides using three different dehydration reagents (i.e. p-toluenesulfonyl chloride (p-TsCl), phosphoryl trichloride (POCl3) and the combination of triphenylphosphane (PPh3) and iodine) were investigated and optimized, while considering the principles of green chemistry. Comparison of the yield and the E-factors of the different synthesis procedures revealed that, in contrast to the typically applied POCl3 or phosgene derivatives, p-TsCl was the reagent of choice for non sterically demanding aliphatic mono- or di-N-formamides (yields up to 98% and lowest E-factor 6.45). Apart from a significantly reduced E-factor, p-TsCl is cheap, offers a simplified reaction protocol and work-up, and is less toxic compared to other dehydration reagents. Thus, this procedure offers easier and greener access to aliphatic isocyanide functionalities.
- This article is part of the themed collection: 2020 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...