Graphene oxide sponge as adsorbent for organic contaminants: comparison with granular activated carbon and influence of water chemistry†
Abstract
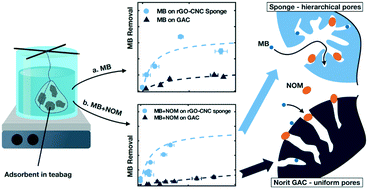
There is growing interest in graphene oxide (GO) as an adsorbent for recalcitrant organic contaminants, such as industrial dyes and pharmaceutical compounds, because of its high specific surface area, amphiphilic character and versatile chemical functionality. GO nanosheets can be self-assembled into three-dimensional porous sponges to facilitate their removal from treated waters. Although a number of chemically and structurally diverse sponges have been fabricated for the removal of anthropogenic water pollutants, adsorption studies have mostly been limited to single contaminant solutions, omitting the environmentally relevant effect of organic co-contaminants and inorganic ions on the adsorption process. Furthermore, the adsorptive performance of the novel material is rarely compared to the industrial standard, granular activated carbon. In this work, we show that a reduced graphene oxide (rGO)–cellulose nanocrystal sponge has a high initial adsorption rate of 17.0 mg g−1 s−1 and the specific adsorption capacity for the model dye methylene blue is three times higher than that of a commonly used commercial granular activated carbon. With a novel HPLC method to quantify methylene blue in the presence of natural organic matter, we show that the adsorption of methylene blue on the sponge is not influenced by environmentally relevant concentrations of natural organic matter. However, organic co-contamination does inhibit the removal of the dye by granular activated carbon. Ionic strength and pH of the contaminant solution do not significantly impact the performance of the rGO sponge. Differences in adsorptive performance between the sponge and GAC are attributed to differences in the surface chemistry and pore structure of the adsorbents.


 Please wait while we load your content...
Please wait while we load your content...