Structure–properties relationship in the hydronium-containing pyrochlores (H3O)1+pSb1+pTe1−pO6 with catalytic activity in the fructose dehydration reaction†
Abstract
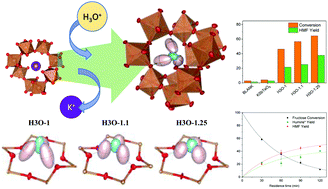
A series of defect pyrochlores of the composition (H3O)1+pSb1+pTe1−pO6 have been prepared by ion exchange from K-containing pyrochlores K1+pSb1+pTe1−pO6 in sulfuric acid at 280 °C for 24 h. The structural characterization of the hydronium-containing pyrochlores, including the location of the H3O+ units within the three-dimensional framework, was possible from neutron powder diffraction data in undeuterated samples. The crystal structure for all the compounds is defined in the Fd![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group, and consists of a covalent framework of SbVO6 and TeVIO6 octahedra distributed at random and connected by their vertices with (Sb,Te)–O1–(Sb,Te) angles close to 136°, conforming to large cages where the hydronium species are located off-center. The absence of K+ ions in the ion-exchanged pyrochlores was confirmed by inductively coupled plasma optical emission spectroscopy and scanning electron microscopy coupled with energy dispersive X-ray spectroscopy. The shape and size of the hydronium units evolve along with the series, becoming more compact as the framework covalence and Lewis-basicity decrease upon Sb enrichment of the structure (for greater p values). The amount and lability of the H3O+ species also increase throughout the series, as wanted: a straightforward correlation of the catalytic activity in the fructose dehydration reaction to 5-hydroxymethylfurfural has been observed, reaching conversion rates up to 88.5% of concentrated fructose solution for the p = 0.25 catalyst. Moreover, a pseudo-first-order kinetic mechanism was simulated, and the kinetic constants obtained from diluted and concentrated enhanced reaction systems were determined and compared.
m space group, and consists of a covalent framework of SbVO6 and TeVIO6 octahedra distributed at random and connected by their vertices with (Sb,Te)–O1–(Sb,Te) angles close to 136°, conforming to large cages where the hydronium species are located off-center. The absence of K+ ions in the ion-exchanged pyrochlores was confirmed by inductively coupled plasma optical emission spectroscopy and scanning electron microscopy coupled with energy dispersive X-ray spectroscopy. The shape and size of the hydronium units evolve along with the series, becoming more compact as the framework covalence and Lewis-basicity decrease upon Sb enrichment of the structure (for greater p values). The amount and lability of the H3O+ species also increase throughout the series, as wanted: a straightforward correlation of the catalytic activity in the fructose dehydration reaction to 5-hydroxymethylfurfural has been observed, reaching conversion rates up to 88.5% of concentrated fructose solution for the p = 0.25 catalyst. Moreover, a pseudo-first-order kinetic mechanism was simulated, and the kinetic constants obtained from diluted and concentrated enhanced reaction systems were determined and compared.


 Please wait while we load your content...
Please wait while we load your content...