Photophysics and ultrafast processes in rhenium(i) diimine dicarbonyls†
Abstract
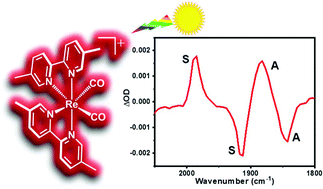
In this work, a series of nine Re(I) diimine dicarbonyl complexes of the general molecular formula cis-[Re(N^N)2(CO)2]+ (N^N are various 2,2′-bipyridine (bpy) or 1,10-phenanthroline (phen) derivatives) were prepared and spectroscopically investigated to systematically evaluate the photophysical consequences of various substituents resident on the diimine ligands. These panchromatic absorbing chromophores were structurally characterized, evaluated for their electrochemical and spectroelectrochemical properties, and investigated using static and dynamic electronic absorption, photoluminescence (PL), and infrared spectroscopy from ultrafast to supra-nanosecond time scales. The ultrafast time-resolved infrared (TRIR) analysis was further supported by electronic structure calculations which characterized the changes within the two C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) O vibrational modes upon formation of the metal-to-ligand charge transfer (MLCT) excited state. The MLCT excited state decay of this series of dicarbonyl molecules appears completely consistent with energy-gap law behavior, where the nonradiative decay rate constants increase logarithmically with decreasing excited state – ground state energy separation, except in anticipated cases where the substituents were phenyl or tert-butyl.
O vibrational modes upon formation of the metal-to-ligand charge transfer (MLCT) excited state. The MLCT excited state decay of this series of dicarbonyl molecules appears completely consistent with energy-gap law behavior, where the nonradiative decay rate constants increase logarithmically with decreasing excited state – ground state energy separation, except in anticipated cases where the substituents were phenyl or tert-butyl.


 Please wait while we load your content...
Please wait while we load your content...