Three metal–organic framework isomers of different pore sizes for selective CO2 adsorption and isomerization studies†
Abstract
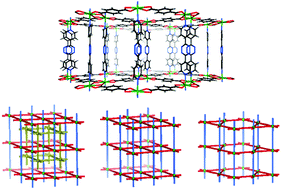
Metal–organic frameworks (MOFs) or porous coordination polymers (PCPs) with tunable pore sizes, shapes and functionalities have excellent prospects in many applications, such as carbon capture. Molecular sieving can usually enable very high CO2 adsorption selectivity but has rarely been achieved, because it is difficult to precisely control the pore size in the range of 3–4 Å. We report here three MOF isomers built from CdII, terephthalic acid and 3,6-di(pyridin-4-yl)-1,2,4,5-tetrazine with the same stoichiometric ratio, among which 1 and 2 are framework-catenation isomers and 2 and 3 are framework-topological isomers. 1 contains 2-fold interpenetrated networks (topology of pcu) and 1D ultra-micropores and shows highly selective adsorption of CO2 over N2 and CH4, which is mainly ascribed to the molecular sieving effect of the framework. 2 contains a pcu network with 3D interconnected micropores, and 3 contains a kag network with much larger pores of 15 Å. Framework isomerization, in this case, was shown to be a feasible way of tuning the pore size of a MOF for selective CO2 adsorption. The effects of hydrothermal reaction conditions and additives on the structures and the formation of the MOF isomers were also studied.


 Please wait while we load your content...
Please wait while we load your content...