Structurally characterized gallium–chrysin complexes with anticancer potential†
Abstract
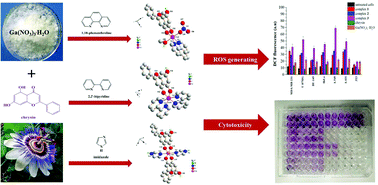
Chemotherapeutic metal-based compounds are effective anticancer agents; however, their cytotoxic profile and significant side effects limit their wide application. Natural products, especially flavonoids, are a prominent alternative source of anticancer agents that can be used as ligands for the generation of new bioactive complexes with metal ions of known biochemical and pharmacological activities. Herein, we present the synthesis and detailed structural and physicochemical characterizations of three novel complex assemblies of Ga(III) with the flavonoid chrysin and the ancillary aromatic chelators 1,10-phenanthroline, 2,2′-bipyridine and imidazole. The complexes constitute the only crystallographically characterized structures having a metal core from the boron group elements and a flavonoid as the ligand. The in vitro biological evaluation of the three complexes in a series of cancer cell lines of different origin established their cytotoxicity and ROS generating potential. In particular, the Ga(III)–chrysin–imidazole complex displayed the highest anticancer efficacy against all cancer cell lines with IC50 values in the low micromolar range (<1.18 μM), a result worth further investigation.
- This article is part of the themed collection: Spotlight Collection focused on Inorganic Chemistry in Greece


 Please wait while we load your content...
Please wait while we load your content...