Seed-assisted hydrothermal synthesis of Sn-Beta for conversion of glucose to methyl lactate: effects of the H2O amount in the gel and crystallization time†
Abstract
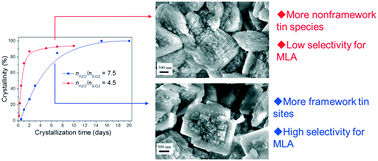
Hydrothermally synthesized Sn-Beta is an excellent catalyst for the conversion of sugars to alkyl lactates. However, its catalytic performance is highly dependent on the synthesis parameters. In this work, the effects of the synthesis parameters such as the water amount of the synthesis gel and the crystallization time on the synthesis and properties and catalytic performance of Sn-Beta in the conversion of glucose to methyl lactate (MLA) were investigated. The crystallization rate was accelerated remarkably by reducing the water amount of the synthesis gel. With the aid of the Si-Beta seed, Sn-Beta was almost fully-crystallized after one day with nH2O/nSiO2 of 4.5, but seven days were needed with nH2O/nSiO2 of 7.5. However, Sn-Beta synthesized with nH2O/nSiO2 of 4.5 has more extraframework SnO2 due to the fast formation of the *BEA structure and the slow incorporation of Sn4+ into the framework sites, which resulted in the lower activity of MLA formation. More Sn4+ species were incorporated into the framework sites and thus more Lewis acid sites were generated with increasing crystallization time. At the same time, the ratio of open to closed Sn sites (RO/C) increased, which led to the decrease in the activity of MLA formation and due to that, the open Sn site with a proximal silanol is unfavorable for the conversion of glucose to MLA. During recycling study, it is surprisingly found that the yield of MLA increased gradually over the fully-crystallized Sn-Beta catalyst. Characterization results of the recovered catalyst from the eighth run indicate that the structure was preserved well, but the value of RO/C reduced due to the change of the state and microenvironment of Sn species.


 Please wait while we load your content...
Please wait while we load your content...