Dehydra-decyclization of tetrahydrofurans to diene monomers over metal oxides†
Abstract
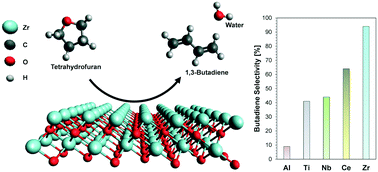
The dehydra-decyclization of tetrahydrofuran (THF) to butadiene was investigated over a series of metal oxide catalysts, where a common set of chemical pathways was identified. Alongside butadiene, propene is formed via a retro-Prins condensation as the main side product typically observed. Reaction occurred at similar temperatures on each of the oxides, but tetragonal zirconia (t-ZrO2) and monoclinic zirconia (m-ZrO2) were unique in showing high selectivity to butadiene (>90%). Near quantitative yields to butadiene could be achieved over t-ZrO2 at 673 K and a WHSV of 0.93 g THF gcat−1 h−1. Through contact time studies, butadiene is determined to be a primary product. Methyl-substituted THF gave only moderate increases in rates and the products showed minimal isomerization of the carbon backbone. The t-ZrO2 catalyst was found to be relatively stable with time on stream, experiencing coking as a likely source of deactivation. Complete regeneration of the catalyst was demonstrated through calcination alone, allowing for multiple regenerations with no irreversible loss in activity or selectivity. The catalytic activity of zirconia was found to be structure insensitive, with t-ZrO2 and m-ZrO2 exhibiting similar initial activities; however, m-ZrO2 was observed to deactivate much more rapidly.


 Please wait while we load your content...
Please wait while we load your content...