Differences in the selective reduction mechanism of 4-nitroacetophenone catalysed by rutile- and anatase-supported ruthenium catalysts†
Abstract
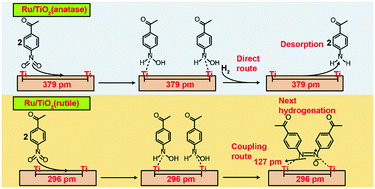
Ru/TiO2 catalysts exhibit excellent catalytic performance for selective reduction of 4-nitroacetophenone to 4-aminoacetophenone at normal temperature and atmospheric hydrogen pressure. Moreover, 99.9% selectivity to 4-aminoacetophenone can be obtained over 2.7 wt% Ru/TiO2(anatase) catalyst even in a relatively wide temperature (55–115 °C) and time (1–12 h) range. Its excellent catalytic performance is derived from the activation of H2 on the Ru nanoparticles at atmospheric pressure and the strong interaction of nitro groups with the support surface. Additionally, Ru nanoparticles supported on different crystalline TiO2 phases (anatase and rutile) result in different reaction pathways for 4-nitroacetophenone. Since the Ti–Ti distance on the rutile surface is smaller than that on the anatase surface, the hydroxylamine species adsorbed on the Ti atoms of rutile are more susceptible to the coupling reaction. Therefore, Ru/TiO2(rutile) causes a series of intermediates to accumulate during the conversion process, while Ru/TiO2(anatase) allows the highly selective conversion of 4-nitroacetophenone to 4-aminophenone. In addition, Ru/TiO2(anatase) can achieve chemoselective reduction of nitroaromatics to the corresponding anilines in the presence of –CN, –CHO, and –COOH, especially nitroaromatics containing C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and C
C and C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C, indicating the excellent applicability.
C, indicating the excellent applicability.


 Please wait while we load your content...
Please wait while we load your content...