Single-molecule kinetic studies of DNA hybridization under extreme pressures†
Abstract
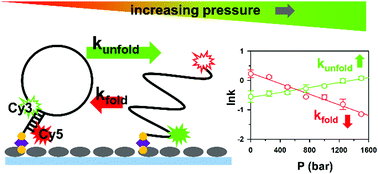
Hydrostatic pressure can perturb biomolecular function by altering equilibrium structures and folding dynamics. Its influences are particularly important to deep sea organisms, as maximum pressures reach ≈1100 bar at the bottom of the ocean as a result of the rapid increase in hydraulic pressure (1 bar every 10 meters) under water. In this work, DNA hybridization kinetics has been studied at the single molecule level with external, tunable pressure control (Pmax ≈ 1500 bar), realized by incorporating a mechanical hydraulic capillary sample cell into a confocal fluorescence microscope. We find that the DNA hairpin construct promotes unfolding (“denatures”) with increasing pressure by simultaneously decelerating and accelerating the unimolecular rate constants for folding and unfolding, respectively. The single molecule kinetics is then investigated via pressure dependent van’t Hoff analysis to infer changes in the thermodynamic molar volume, which unambiguously reveals that the effective DNA plus solvent volume increases (ΔV0 > 0) along the folding coordinate. Cation effects on the pressure dependent kinetics are also explored as a function of monovalent [Na+]. In addition to stabilizing the overall DNA secondary structure, sodium ions at low concentrations are also found to weaken any pressure dependence for the folding kinetics, but with these effects quickly saturating at physiologically relevant levels of [Na+]. In particular, the magnitudes of the activation volumes for the DNA dehybridization (ΔV‡unfold) are significantly reduced with increasing [Na+], suggesting that sodium cations help DNA adopt a more fold-like transition state configuration.
- This article is part of the themed collection: 2020 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...