Borophosphene as a promising Dirac anode with large capacity and high-rate capability for sodium-ion batteries†
Abstract
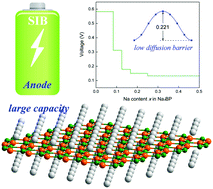
Sodium-ion batteries (SIBs) have attracted tremendous attention as potential low-cost energy storage alternatives to lithium-ion batteries (LIBs) due to the intrinsic safety and great abundance of sodium. For developing competitive SIBs, highly efficient anode materials with large capacity and rapid ion diffusion are indispensable. In this study, a two-dimensional (2D) Dirac monolayer, that is, borophosphene, is proposed as a promising anode material for high performance SIBs on the basis of density functional theory calculations. The performances of Na adsorption and diffusion, maximum specific capacity, open circuit voltage, cyclical stability and electronic properties combined with Bader charge analysis are explored. It is found that borophosphene can spontaneously adsorb a Na atom with a binding energy of −0.838 eV. A low diffusion energy barrier of 0.221 eV suggests rapid ion conductivity. More intriguingly, a maximum specific capacity of 1282 mA h g−1 can be achieved in borophosphene, which is one of the largest values reported for 2D anode materials for SIBs. A low average voltage of 0.367 V is estimated, implying a suitable operating voltage of the anode material. The metallic properties, tiny surface expansion, and good kinetic stability of sodiated borophosphene give rise to high electrical conductivity and favorable cyclability. These abovementioned advantages suggest that borophosphene can be used as a Dirac anode material for SIBs with excellent performance including a large specific capacity, high-rate capability, and favorable cyclability.


 Please wait while we load your content...
Please wait while we load your content...