IR–VUV spectroscopy of pyridine dimers, trimers and pyridine–ammonia complexes in a supersonic jet†
Abstract
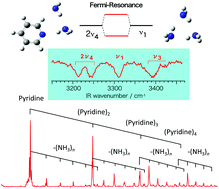
The infrared spectra of the C–H stretching vibrations of (pyridine)m, m = 1–3, and the N–H stretching vibrations of (pyridine)m–(NH3)n, m = 1, 2; n = 1–4, complexes were investigated by infrared (IR)–vacuum ultraviolet (VUV) spectroscopy under jet-cooled conditions. The ionization potential (IP0) of the pyridine monomer was determined to be 74 546 cm−1 (9.242 eV), while its complexes showed only smooth curves of the ionization thresholds at ∼9 eV, indicating large structural changes in the ionic form. The pyridine monomer exhibits five main features with several satellite bands in the C–H stretching region at 3000–3200 cm−1. Anharmonic calculations including Fermi-resonance were carried out to analyze the candidates of the overtone and combination bands which can couple to the C–H stretching fundamentals. For (pyridine)2 and (pyridine)3, most C–H bands are blue-shifted by 3–5 cm−1 from those of the monomer. The structures revealed by random searching algorithms with density functional methods indicate that the π-stacked structure is most stable for (pyridine)2, while (pyridine)3 prefers the structures stabilized by dipole–dipole and C–H⋯π interactions. For the (pyridine)m–(NH3)n complexes, the mass spectrum exhibited a wide range distribution of the complexes. The observed IR spectra in the N–H stretching vibrations of the complexes showed four main bands in the 3200–3450 cm−1 region. These features are very similar to those of (NH3)n complexes, and the bands are assigned to the anti-symmetric N–H stretching band (ν3), the symmetric N–H stretching (ν1) band, and the first overtone bands of the N–H bending vibrations (2ν4). The anharmonic calculations including the Fermi-resonance between ν1 and 2ν4 well reproduced the observed spectra.


 Please wait while we load your content...
Please wait while we load your content...