Achieving tunable emitting YAl3(BO3)4:Eu phosphors by substituting cations (Sr2+, Ca2+, Ga3+, In3+) and controlling BO3 group polarizability
Abstract
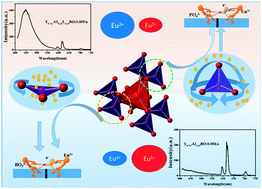
In order to explore the influence factor on the luminescence characteristics and valence state of Eu ions, a series of YAl3(BO3)4−m(PO4)m:0.05Eu, Y0.925−xACxLxBO:0.05Eu, Y0.925−yASyLyBO:0.05Eu, Y0.925−zAGzBO:0.05Eu and Y0.925−wAIwBO:0.05Eu phosphors are synthesized in a CO atmosphere. It is found that the content ratio of Eu2+ to Eu3+ ions is closely related to the crystal volume. The larger the volume, the larger the polarizability of BO3 anionic groups, and the larger the content ratio of Eu2+ to Eu3+ ions. When Y3+ cations in YAl3(BO3)4 are replaced by Sr2+ or Ca2+ ions with a larger radius, the ratio of Eu2+ to Eu3+ ions increases, and the maximum ratio is 0.798. When Ga3+ ions with a radius smaller than that of Y3+ ions but larger than that of Al3+ ions are doped, Al3+ ions are preferentially replaced by Ga3+ ions, resulting in the increase of crystal volume, and the ratio of Eu2+ ions to Eu3+ ions increases from 0.159 to 0.359. When In3+ ions with a radius smaller than that of Y3+ ions but larger than that of Al3+ ions are added, the crystal volume decreases with increasing In3+ concentration, and the proportion of Eu2+ to Eu3+ ions decreases accordingly; moreover, new luminescence centers are observed, which come from oxygen vacancy defects. In short, the results indicate that the luminescence properties, especially the content ratio of Eu2+ to Eu3+, can be adjusted by regulating the matrix components, and this should be used to adjust the white emission or red–blue ratio.


 Please wait while we load your content...
Please wait while we load your content...