Polymorph induced diversity of photomechanical motions of molecular crystals†
Abstract
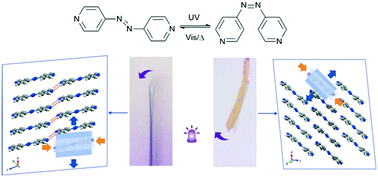
Although diverse molecule structures have been designed to realize photoresponsive motion, the effects of polymorphism on photoresponsiveness are rarely reported. Here, we utilize trans-4,4′-azopyridine (trans-4AP) as a model compound to investigate the effect of crystal structures on photomechanical motion. A new polymorph of the trans-4AP crystal, form 2, was discovered and compared with the known form 1 from the perspective of molecular arrangement and intermolecular interactions. It was found that different molecular arrangements resulted in distinct performance of photoisomerization in the two types of solids. Under UV light, the form 1 crystals bent away from the UV light, while the form 2 crystals exhibited diverse photomechanical motions including quasi-bidirectional bending and twisting motion, depending on the different sizes and shapes of the used crystals. Such deformation originated from the diverse strain distribution in the irradiated surfaces of form 1 and form 2, which was verified by powder X-ray diffraction, solid-state UV/vis absorption spectroscopy and atomic force microscopy (AFM). Various contributions and energies of intermolecular interactions make the two polymorphs of trans-4AP crystals deform in different modes. Relatively weak intermolecular interaction resulted in larger deformation in form 2, which was further proved by the Hirshfeld surface and energy framework analysis.


 Please wait while we load your content...
Please wait while we load your content...