A gold(i)-catalysed chemoselective three-component reaction between phenols, α-diazocarbonyl compounds and allenamides†
Abstract
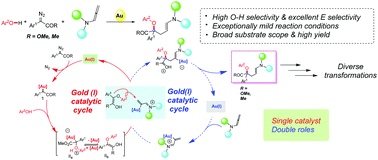
Gold(I) catalysts are considered to be efficient in promoting sp2 C–H bond insertion at the para position of unprotected phenols because of the exceptionally chemical reactivity of gold-stabilized carbophilic carbocations. Herein, we present a gold(I)-catalysed three-component reaction between phenols, diazocarbonyl compounds and allenamides, affording the corresponding three-component reaction products with excellent geometric selectivity (E : Z > 20 : 1) in moderate to high yields (up to 90%) under mild conditions. Additionally, a gram-scale transformation and diverse transformations of the resulting product show the high synthetic utility of the present three-component protocol.


 Please wait while we load your content...
Please wait while we load your content...