Aquaporin-driven hydrogen peroxide transport: a case of molecular mimicry?†
Abstract
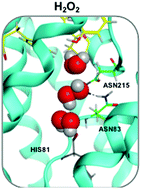
Aquaporins (AQPs) are membrane proteins that have evolved to control cellular water uptake and efflux, and as such are amongst the most ancient biological “devices” in cellular organisms. Recently, using metadynamics, we have shown that water nanoconfinement within aquaporin channels results into bidirectional water movement along single file chains, extending previous investigations. Here, the elusive mechanisms of H2O2 facilitated transport by the human ‘peroxiporin’ AQP3 has been unravelled via a combination of atomistic simulations, showing that while hydrogen peroxide is able to mimic water during AQP3 permeation, this comes at a certain energy expense due to the required conformational changes within the channel. Furthermore, the intrinsic water dynamics allows for host H2O2 molecule solvation and transport in both directions, highlighting the fundamental role of water nanoconfinement for successful transduction and molecular selection. Overall, the bidirectional nature of the water flux under equilibrium conditions along with the mimicking behavior of hydrogen peroxide during a conductance event introduce a new chemical paradigm never reported so far in any theoretical paper involving any aquaporin isoform.
- This article is part of the themed collection: Exploring proteins and their interactions


 Please wait while we load your content...
Please wait while we load your content...