An ionic-based carbon dot for enantioselective discrimination of nonaromatic amino alcohols†
Abstract
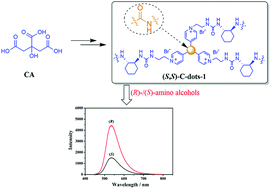
Here, ionized chiral carbon dots, (S,S)-C-dots-1 (λex = 430 nm, λem = 480 nm), were synthesized via a facile route with relatively high quantum yield (∼24.4%) and used as a fluorescent chiral sensor. One of the advantages of the synthetic process is that it avoids the loss of the chiral center. That is, the chiral bromo compound can directly form an ionic pair with the pyridyl group, which is derived from the amine precursor in the first step. Furthermore, (S,S)-C-dots-1 shows clear discrimination toward different configurations of nonaromatic amino alcohols in the presence of Cu(II). When the (R)-isomer is added to a solution of (S,S)-C-dots-1 + Cu(II), it shows much higher fluorescent intensity than the (S)-isomer. The values of IR/IS are 2.9 and 2.3 for 2-aminobutan-1-ol and 2-aminopropan-1-ol, respectively. In summary, we believe that this work can expand the synthetic routes and potential applications of functional carbon dots in the field of enantioselective sensing.


 Please wait while we load your content...
Please wait while we load your content...