Versatile antifouling coatings based on self-assembled oligopeptides for engineering and biological materials†
Abstract
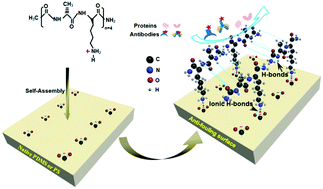
The existence of nonspecific protein adsorption often results in significant challenges for microfluidic devices and laboratory cultureware used in biological experiments. Developing antifouling surfaces is thus increasingly desired by microfluidics engineers and biologists. Our previous studies have demonstrated that ionic hydrogen bonding between free ε-NH2 groups of the alkaline amino acids in proteins and surface negatively charged groups plays a critical role in strong adsorption of proteins onto the solid surfaces. Thus, the current work presents a facile and universal surface modification method based on self-assembly of oligopeptides with a sequence of Ala-Lys-Ala-Lys-Ala-Lys-Ala-Lys (AK-VIII) on poly(dimethylsiloxane) (PDMS) and polystyrene (PS) surfaces under physiological pH conditions. The results show that AK-VIII can self-organize into a compact amphipathic β-sheet-rich coating layer on the PDMS and PS surfaces with similar coverage, which largely minimizes nonspecific adsorption of proteins. In addition, we compared the performances of BSA and AK-VIII used as blocking reagents to evaluate their inhibitory effect on nonspecific adsorption of the antigen and detection antibody in carcinoembryonic antigen (CEA) enzyme-linked immunosorbent assay (ELISA). The results showed that AK-VIII exhibits better performance than BSA for diminishing nonspecific adsorption of the antigen and detection antibody, thus providing lower background noise, a lower detection limit, and a wider linear range in CEA assays. This study provides a novel and versatile alternative for developing antifouling coatings to address nonspecific protein adsorption on both PDMS-based engineering and PS-based biological materials.
- This article is part of the themed collection: 2019 Journal of Materials Chemistry B HOT Papers


 Please wait while we load your content...
Please wait while we load your content...