Fluorescent silicon nanoparticles inhibit the amyloid fibrillation of insulin†
Abstract
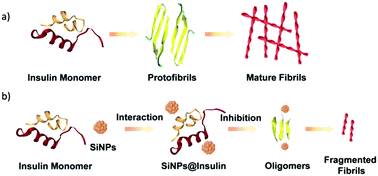
Amyloid fibrillation of proteins is likely a key factor leading to the development of amyloidosis-associated diseases. Inhibiting amyloid fibrillation has become a crucial therapeutic strategy. Water-soluble, fluorescent silicon nanoparticles (SiNPs) have great potential in biomedicine for various therapeutic and diagnostic purposes; however, it is unclear whether SiNPs have the ability to inhibit amyloid fibrillation. Herein, insulin was chosen as a protein model, and SiNPs of varying sizes were synthesized upon UV irradiation. The influence of size and concentration of the SiNPs on insulin fibrillation was investigated, and it has been observed that these variables were crucial in regulating insulin fibrillation. Using an average particle size of 6.6 nm and increasing the concentration of the SiNPs to 5.0 μg mL−1, the Thioflavin T (ThT) fluorescence intensity decreased significantly by 90%, with an increased lag time of 76.8 h, compared to that of the control. Insulin aggregates were short, thin fibrils or clusters when incubated with SiNPs, compared to the long, thick fibrils formed for insulin alone. Additionally, we found that the SiNPs prevent the conformational transition of insulin from its initial structure to β-sheets, and thus inhibit nucleation, which is necessary for the formation of large fibrils. The inhibitory activity is attributed to the interactions between the SiNPs and insulin during the nucleation period. Our results demonstrate that the SiNPs disrupt insulin amyloid fibrillation, and thus, may play a useful role in new therapeutic and diagnostic strategies for amyloid-related disorders.


 Please wait while we load your content...
Please wait while we load your content...