A high-performance cupric oxide photocatalyst with palladium light trapping nanostructures and a hole transporting layer for photoelectrochemical hydrogen evolution†
Abstract
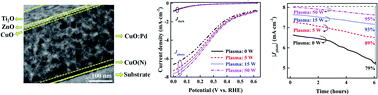
The high recombination rate of photogenerated electron–hole pairs, poor photocorrosion stability, and the discrepancy between the optical absorption length and charge collection efficiency of cupric oxide (CuO) are the main limiting factors of visible-light-driven CuO photocatalysts for hydrogen evolution and photocatalytic degradation of organic pollutants. In this paper, we demonstrate a novel thin CuO film photocatalyst on a fluorine doped tin oxide (FTO) coated glass substrate with low back contact resistivity, high charge collection efficiency, high optical absorption, and high photocorrosion stability for hydrogen production and photocatalytic degradation of organic pollutants. This photocatalyst was fabricated by incorporating palladium (Pd) nanostructures into CuO to form a CuO:Pd light trapping thin film. The CuO:Pd light trapping thin film was then sandwiched between a nitrogen doped cupric oxide [CuO(N)] hole transporting layer and a CuO capping layer [CuO(N)–CuO:Pd–CuO]. The performance of the CuO(N)–CuO:Pd–CuO photocatalyst is further improved by incorporating a ZnO buffer layer and TiO2 protective layer, and decorating with a AuPd co-catalyst. Moreover, we demonstrate a significant improvement of photocorrosion stability and photocatalytic degradation efficiency of the CuO(N)–CuO:Pd–CuO–ZnO–TiO2 photocatalyst through plasma assisted in situ nano-crystal engineering of the ZnO buffer layer and TiO2 protective layer. The fabricated novel photocatalyst could retain 95% of the initial photocurrent density after 6 hours of standard illumination with solar light and could give a record high photocurrent density of ∼8 mA cm−2 for the CuO photocatalyst.


 Please wait while we load your content...
Please wait while we load your content...