Superior electrocatalytic hydrogen evolution at engineered non-stoichiometric two-dimensional transition metal dichalcogenide edges†
Abstract
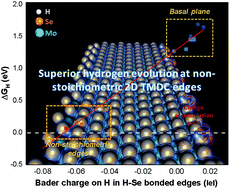
Two-dimensional transition metal dichalcogenide (TMDC) edges show activity for the catalytic hydrogen evolution reaction (HER), but further improvements require extrinsic doping, usually performed in an Edisonian manner. Herein we investigate if tuning the non-stoichiometric degree of the edges itself can improve HER activities. Using first-principles density functional theory (DFT), we study six non-stoichiometric MoSe2 edges that have been recently synthesized under a scanning transmission electron microscope (STEM). We find that non-stoichiometric edges can have near optimal HER activity over conventional stoichiometric edges. More excitingly, we find a strong linear correlation between Bader charges on H and the Gibbs free energy of hydrogen adsorption (ΔGH) at these edges, providing a design principle for discovering better HER catalytic edges. In general, HER activity is not only influenced by the formation of H–Se/Mo chemical bonds as previously thought, but also by geometric reconstructions and charge redistribution. Our predictions open the door for engineering non-stoichiometric TMDC edges for superior HER activity.


 Please wait while we load your content...
Please wait while we load your content...