Synthesis of highly effective stabilized CaO sorbents via a sacrificial N-doped carbon nanosheet template
Abstract
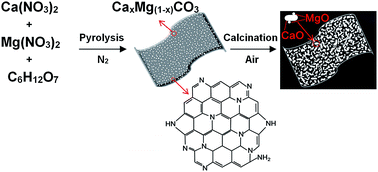
Calcium looping, a promising high-temperature CO2 capture technique, offers a mid-term economic solution to mitigate anthropogenic CO2 emissions. The main challenge in calcium looping is the synthesis of highly efficient CaO-based sorbents that can be used over many reaction cycles. Here, a sacrificial N-doped carbon nanosheet template was developed which produces MgO-stabilized CaO sorbents with fast adsorption rates, high capacities and remarkable long-term performance over many cycles. The characterization results show that such a template was formed through in situ pyrolysis of an organic acid and nitrates in a simple heating process in nitrogen. The presence of a carbonaceous template prevented crystallite growth, featured highly macroporous nanosheet (∼60 nm thick) morphologies, and ensured homogeneous mixing of Ca and Mg, which are essential to minimize diffusion limitations, mitigate sintering, and provide structural stability. Thus, 10 mol% MgO acting as an inert stabilizer was sufficient to achieve a CO2 uptake of 0.65 g g−1 (corresponding to a capacity retention of 89.9%) after 10 cycles under realistic conditions, as confirmed by TGA analysis. This N-doped carbon template can be applied generally to produce a wide range of porous and nanostructured stabilized CaO sorbents with stable CO2 uptake.


 Please wait while we load your content...
Please wait while we load your content...