The importance of morphology on the activity of lead cathodes for the reduction of carbon dioxide to formate†
Abstract
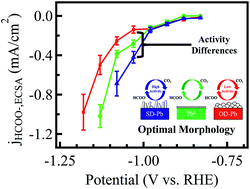
The selectivity of carbon dioxide (CO2) electroreduction to formate on lead (Pb) catalysts can be improved by using oxide-derived (OD) materials. However, the activity on these materials remains low. In this work, we unexpectedly discovered that the formate-producing activity of Pb materials is correlated to their morphologies. We have prepared sulfide-derived (SD)-Pb and OD-Pb electrodes by respectively electroreducing PbS and PbOx precursors. SD-Pb had a morphology consisting of wafer structures oriented edge-on, while OD-Pb had more contiguous, rounded features. The SD-Pb cathodes reduced CO2 to formate (HCOO−) with good selectivity and activity in 0.1 M KHCO3 electrolyte (Faradaic efficiency = 88% and geometric partial current density, jHCOO−,geom = −12 mA cm−2 at −1.08 V vs. RHE). When normalized to the electrochemically active surface area (ECSA), the jHCOO−,ECSA of the SD-Pb was distinctly larger than those of OD-Pb and polished Pb discs. X-ray photoelectron spectroscopy (XPS) showed no detectable trace of sulfur on the SD-Pb, and Tafel analysis did not suggest any mechanistic differences between the reactivity of SD-Pb, OD-Pb and Pb. These analyses ruled out chemical and mechanistic explanations for the increased activity of SD-Pb, and indicated that morphological differences are the reason for the variation in activity. Extensive control experiments with Pb surfaces prepared in different ways were performed. We provide compelling evidence showing that only Pb samples with the wafer structures exhibited enhanced selectivity and jHCOO−,ECSA, even when no sulfur was used in the preparation method. Our work demonstrates, for the first time, that the morphology of the Pb cathodes is a critical parameter that influences their catalysis of CO2 reduction to formate.


 Please wait while we load your content...
Please wait while we load your content...