Pt-rare earth metal alloy/metal oxide catalysts for oxygen reduction and alcohol oxidation reactions: an overview
Abstract
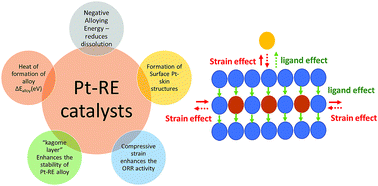
Pt based catalysts represent the state-of-the-art electrocatalyst for electrochemical cathodic oxygen reduction reaction of proton exchange membrane fuel cells. Among Pt based electrocatalysts, Pt-transition metal catalysts have been the most successful in terms of activity. However, the poor structural and chemical stability of Pt-alloy catalysts are challenges. Alternatively, alloying Pt with rare earth metals is considered as one of the effective ways to discover active and durable electrocatalysts for the ORR. The advantages of alloying with rare earth metals bring a substantial change in the electronic properties and their negative alloying energy or enthalpy of formation of alloys provides special driving force to resist against dissolution. Synthesis of Pt-rare earth alloys by a solution phase route is challenging. To date there have been only physical metallurgical methods which help to synthesize them successfully in the form of an alloy, where as traditional wet chemical synthesis methods results rare earth turns into metal oxide/hydroxide. This article reviews several studies put forward to synthesize Pt-rare earth alloy/oxide catalysts by various physical metallurgical methods and solution-based synthesis protocols and their effect on the improvement of ORR activity.


 Please wait while we load your content...
Please wait while we load your content...